Our programs
Discover the future therapeutic solutions of chronic rhinitis and chronic rhinosinusitis - The Dianosic Active Resorbable Intranasal Scaffolds (DIAXX).
Dianosic develops the innovative DIAXX platforms that benefit from proprietary drug-eluting, long-term resorbable polymer.

There a is a need for treatment paradigm change to better manage patients, optimize treatment efficacy and safety.
85 % patients suffering from chronic rhinitis do not comply with their treatment.⁷
20 % patients suffering from chronic rhinosinusitis do not respond to intranasal corticosteroids.⁸
Only 20% of nasal spray active ingredients reach the targeted treatment area.⁹
Dianosic unique platform - Created to radically transform the management of these debilitating diseases
Provide patient-centered “insert-and-forget” solutions that maximize patients’ quality of life while meeting physicians’ needs in terms of efficiency, safety, ease of use, and patient adherence to treatment.

A unique, long-term bioresorbable product architecture designed to optimally deliver a low, targeted dose of effective drug therapy in a controlled and stable manner.
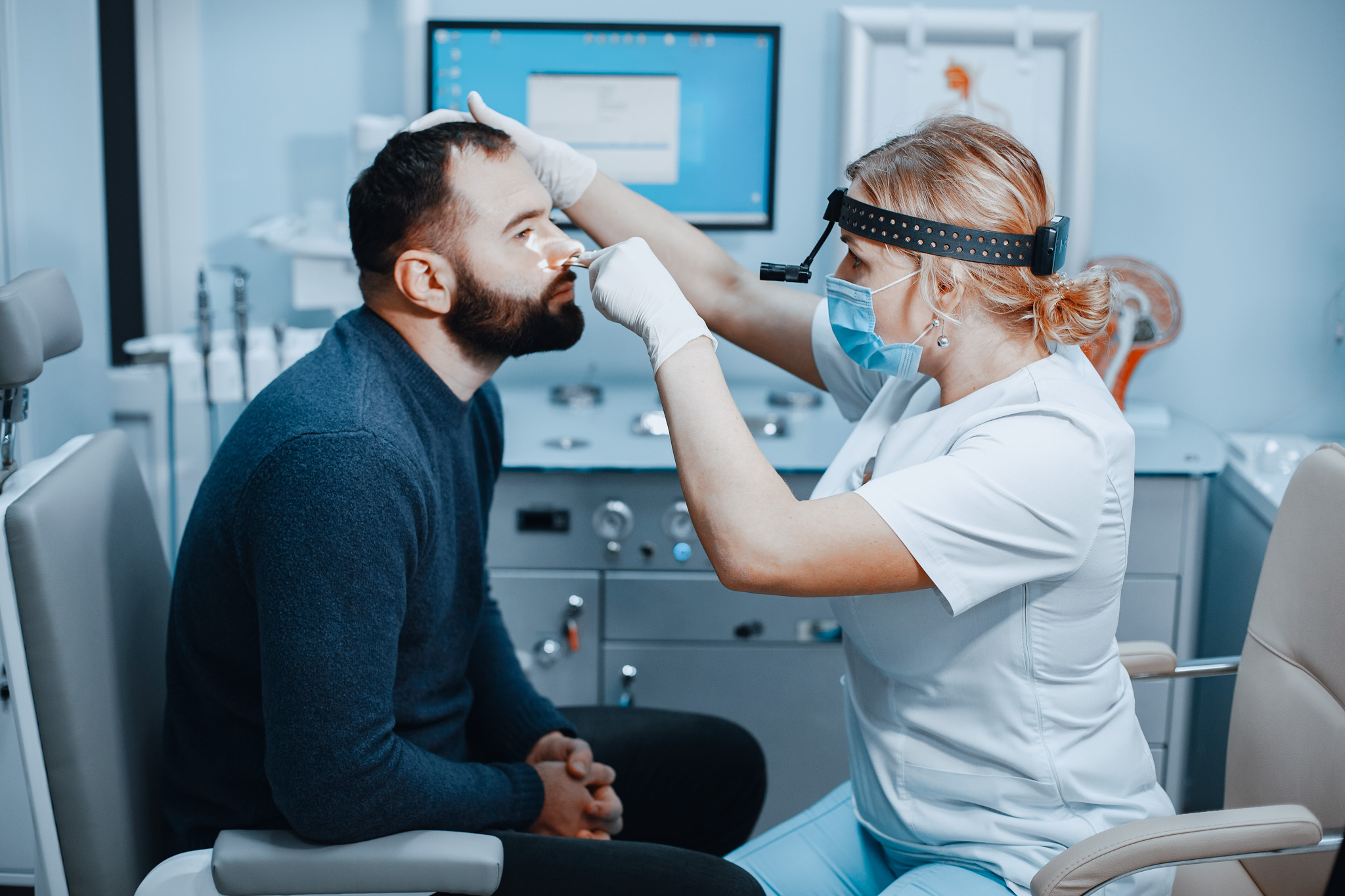
“In-office” implantation under local anesthesia, generating significant cost savings for healthcare systems.

100% treatment adherence (compared to 15% for nasal sprays in rhinitis¹⁰) thanks to our “insert-and-forget” solution.

Unique scaffold architecture combined with proprietary drug-eluting, long-term resorbable polymer
Our DIAXX platform was developed to treat both chronic rhinitis (DIA01-R) and chronic rhinosinusitis (DIA02-S), and differs fundamentally from existing solutions (bioresorbable structure, “insert-and-forget” approach, treatment duration, targeted delivery, low dose, sustained drug release).

DIA01-R
Chronic rhinitis
DIA01-R is the ideal solution for controlling symptoms of moderate-to-severe persistent allergic rhinitis. It delivers a low dose of corticosteroid locally over 6 months. DIA01-R provides an alternative to intranasal corticosteroids and more invasive treatments, while improving treatment adherence and helping prevent symptom worsening.

DIA02-S
Chronic rhinosinusitis
DIA02-S is adapted to long-term symptomatic treatment of chronic rhinosinusitis in adults. It is placed into the nasal cavity under local anesthesia during a less than 30 minutes in-office procedure. DIA02-S delivers locally a low dose or corticosteroids for 12 months offering long-term symptoms relief for patients.

DIA03-NTB Solution
The NTB (Nose To Brain) program explores the significant therapeutic potential of using the intranasal route for drug delivery directly to the brain. This innovative approach targets critical needs in treating central nervous system diseases for which existing treatments face major compliance challenges and can generate significant side effects due to systemic exposure.
The NTB program leverages Dianosic’s expertise in the intranasal route. This approach is based on our know-how in drug eluting polymers acquired with the DIA01-R scaffold development in Chronic Allergic Rhinitis, making it transposable to the development of solutions to treat central nervous system diseases (e.g. Schizophrenia and Parkinson).

Watch Pr. Wytske
Fokkens' video
Click on the button below to watch Pr. Wytske
Fokkens’ video regarding our DIAXX solutions.
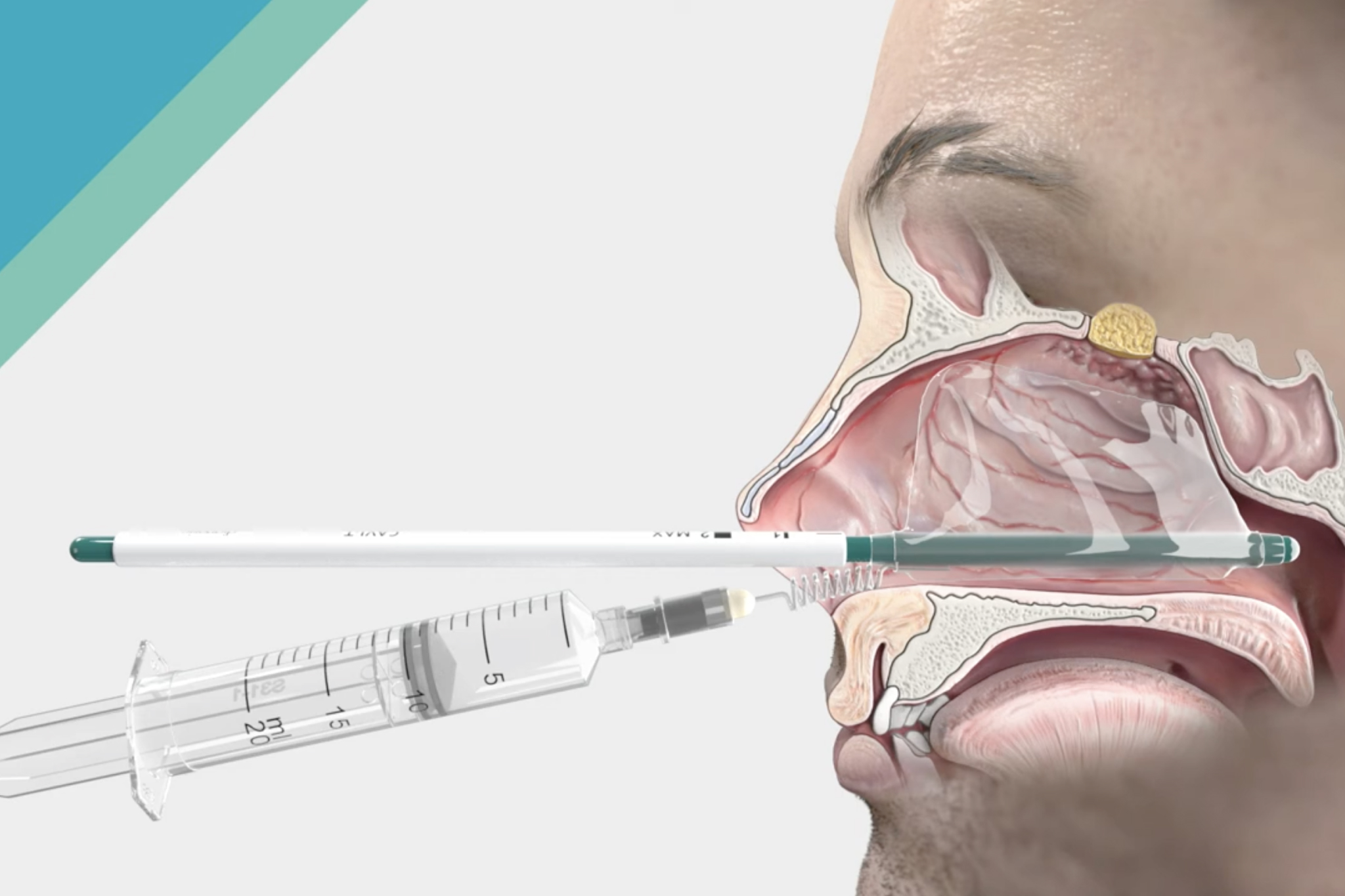
Discover our new solution for epistaxis – CAVI-T
Dianosic successfully developed and launched a versatile and easy to use solution designed to transform the management of intranasal bleeding (epistaxis). CAVI-T illustrates our continuous commitment to deliver innovative solutions to the ENT community.
³ Prevalence and Incidence of Diagnosed Chronic Rhinosinusitis in Alberta, Canada. Yuan Xu et al. JAMA Otolaryngol Head Neck Surg. 2016;142(11):1063-1069. doi:10.1001/jamaoto.2016.2227
⁴ Bauchau, V., & Durham, S. R. (2005). Epidemiological characterization of the intermittent and persistent types of allergic rhinitis. Allergy, 60(3), 350–353.doi:10.1111/j.1398-9995.2005.00751.x
⁵ Antonicelli L, Micucci C, Voltolini S, Feliziani V, Senna GE, Di Blasi P, et al. Allergic rhinitis and asthma comorbidity: ARIA classification of rhinitis does not correlate with the prevalence of asthma. Clin Exp Allergy. 2007 Jun;37(6):954-60.
⁶ Erkkinen MG, Kim MO, Geschwind MD. Clinical Neurology and Epidemiology of the Major Neurodegenerative Diseases. Cold Spring Harb Perspect Biol. 2018 Apr 2;10(4):a033118. doi: 10.1101/cshperspect.a033118. PMID: 28716886; PMCID: PMC5880171.Based on KOLs feedback, we estimate that the % acceptance for the technology is +/-60% — Velligan DI, Rao S. The epidemiology and global burden of schizophrenia. J Clin Psychiatry. 2023;84(1):MS21078COM5. Based on KOLs feedback, we estimate that the % acceptance for the technology is +/-50%
⁷⁻¹⁰ Menditto E et al. Adherence to treatment in allergic rhinitis using mobile technology. The MASK Study. Clin Exp Allergy. 2019 Apr;49(4):442-460. doi: 10.1111/cea.13333.
⁸ The Role of Balloon Sinuplasty in the Treatment of Chronic Sinusitis. Cummings JP et al. JCOM. 2009; 16(1):30-6.
⁹ La dynamique des fluides numérique (CFD) a été utilisée pour étudier les profils de dépôt des particules issues de dispositifs de pulvérisation [ 12 – 19 ]. Ces études ont révélé de faibles efficacités de dépôt dans la partie moyenne et postérieure de la cavité nasale et dans les sinus, où l’absorption se produit.











